Category: Medications - Page 2
Learn about medication-induced Acute Angle-Closure Glaucoma, a severe eye emergency. Discover high-risk drugs, warning signs, and how to prevent permanent vision loss.
Learn how to verify your name and medication on a prescription label to prevent errors. This guide covers legal requirements, verification steps, and tools for patient safety.
Explore the history of thalidomide, the medical tragedy that reshaped drug safety laws, and how it is used cautiously today for cancer treatment.
Explore what bioequivalence testing really proves regarding generic versus brand-name drugs. Learn how regulatory standards ensure safety and efficacy.
Counterfeit drugs in developing nations kill tens of thousands annually, with fake medicines containing no active ingredients or toxic chemicals. Learn how they spread, why they're so dangerous, and what's being done to stop them.
Doctors need ongoing education on generic medications to prescribe safely, improve adherence, and reduce costs. With 90% of prescriptions now generic, CME must evolve beyond checklists to real-world, patient-centered learning.
Warfarin users must avoid common OTC cold medications like ibuprofen and aspirin to prevent dangerous bleeding. Learn which ingredients are safe, how to read labels, and what to do when you're sick - backed by clinical data and real patient stories.
FDA inspections of generic drug manufacturing facilities are routine but rigorous. Learn what inspectors look for, how to prepare, and what happens if issues are found-all based on current 2026 regulations and real-world practices.
Therapeutic drug monitoring helps protect patients on generic HIV drugs by measuring blood levels to prevent toxicity and treatment failure-especially for protease inhibitors and NNRTIs. Not for all drugs, but critical in high-risk cases.
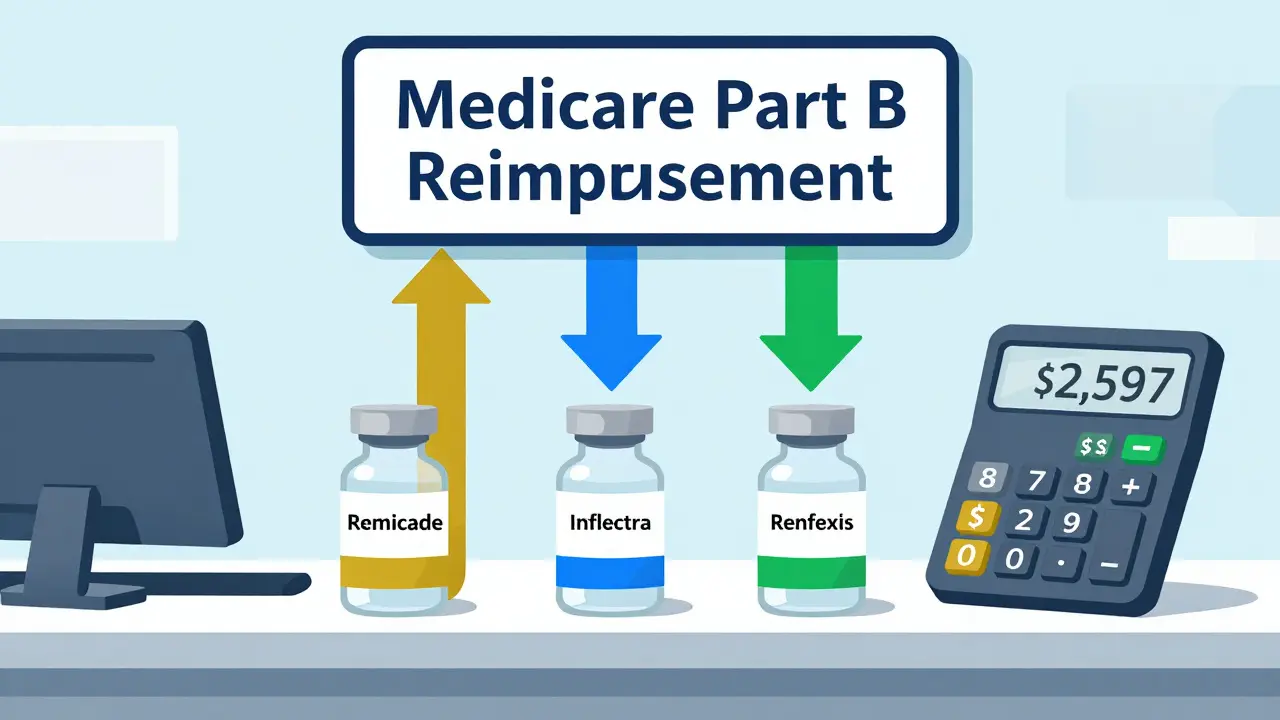
Biosimilars are billed differently than generics under Medicare Part B. Each has its own HCPCS code and payment rate based on its ASP plus 6% of the reference product’s price. This system encourages biosimilar entry but doesn’t strongly incentivize switching. Learn how billing works and why adoption remains low in the U.S.










 Medications
Medications




