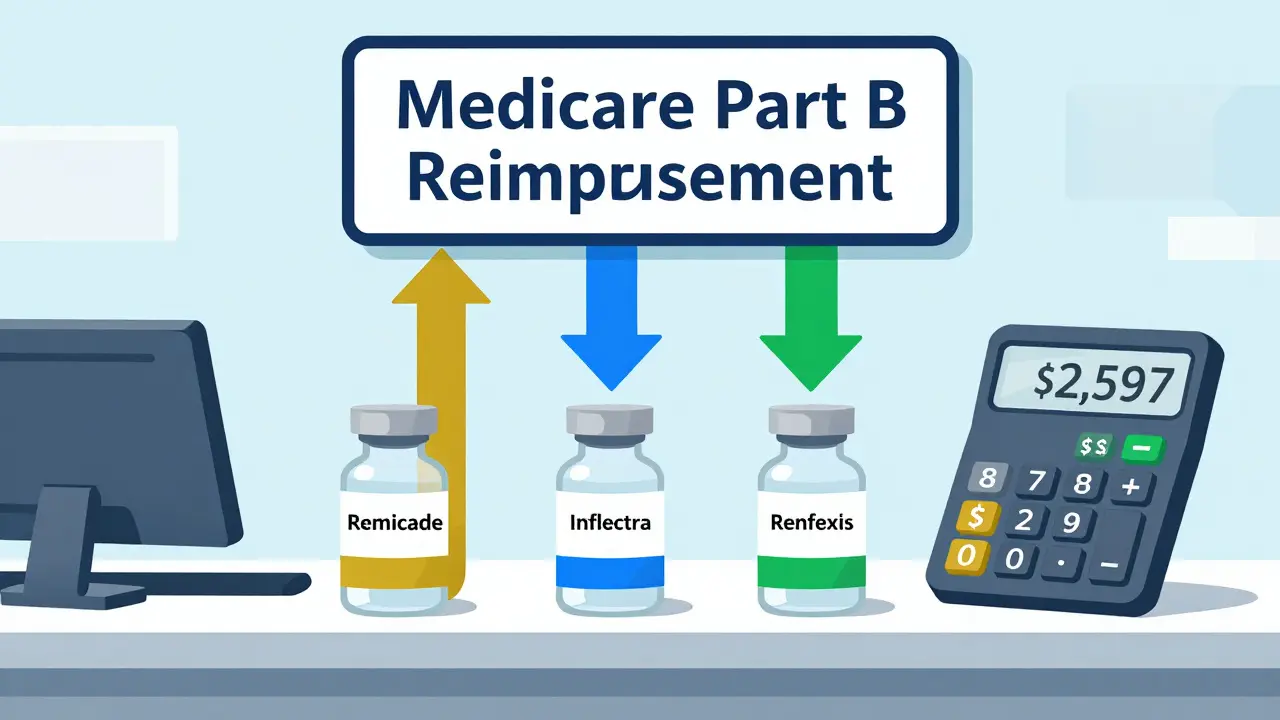
When a patient receives a biosimilar drug like Inflectra or Renflexis instead of the original biologic Remicade, the billing process isn’t as simple as switching one pill for another. Unlike generic drugs, which are chemically identical and use the same code for all brands, biosimilars are complex biological products that each get their own billing code and payment rate. This makes reimbursement for biosimilars one of the most confusing parts of modern healthcare billing - especially under Medicare Part B, which covers drugs given in clinics and doctor’s offices.
How Biosimilars Are Coded Differently Than Generics
Generic drugs, like metformin or lisinopril, are treated as interchangeable. Once a brand-name drug loses patent protection, any manufacturer can make the same chemical compound and use the same HCPCS code - J0151 for metformin, for example. But biosimilars? They’re not generics. Even though they’re highly similar to the original biologic, they’re not identical. That’s why the Centers for Medicare & Medicaid Services (CMS) stopped using a single shared code for all biosimilars of the same drug after 2017.
Before January 2018, all infliximab biosimilars - like Inflectra, Renflexis, and Ixifi - were billed under one code: Q5101. That meant if Inflectra sold for $2,000 per dose and Renflexis sold for $1,800, providers got paid the same blended rate for both. The problem? It created a disincentive for manufacturers to enter the market with lower-priced options. Why bother if your lower cost doesn’t mean higher reimbursement?
In 2018, CMS flipped the script. Each biosimilar now has its own unique HCPCS code - either a temporary Q-code or a permanent J-code. Inflectra got J3590, Renflexis got J3591, and so on. This change forced payment to match the actual price of each product. Now, if Inflectra costs $2,000, you get paid 100% of $2,000 plus 6% of the reference product’s price (Remicade). If Renflexis costs $1,800, you get paid 100% of $1,800 plus 6% of Remicade’s price. The difference in your reimbursement? It’s real, and it’s based on what you actually gave the patient.
The 106% Payment Rule - What It Really Means
Medicare Part B pays for most physician-administered drugs at 106% of the Average Selling Price (ASP). That means 100% for the drug itself, plus a 6% add-on to cover handling, storage, and administration costs. For biosimilars, CMS calculates this add-on using the ASP of the reference product, not the biosimilar.
Let’s say Remicade (the original) sells for $2,500 per dose. Its ASP is $2,450. Inflectra sells for $2,000, with an ASP of $1,950. Under the current rule, here’s what a provider gets paid:
- For Remicade: $2,450 (ASP) + 6% of $2,450 = $2,597
- For Inflectra: $1,950 (ASP) + 6% of $2,450 = $2,097
That’s a $500 difference in reimbursement - even though Inflectra is 20% cheaper. The provider earns $150 in add-on revenue from Remicade but only $147 from Inflectra. The math doesn’t favor switching. Critics call this a perverse incentive. Why would a clinic push a cheaper drug if it means losing $3 per dose in revenue? Dr. Mark Trusheim from MIT calculated this exact gap in 2020 and found that for high-cost biologics, providers actually make more money by sticking with the brand-name drug.
The JZ Modifier and the New Documentation Burden
In July 2023, CMS added another layer: the JZ modifier. If a provider administers a drug like infliximab and discards zero doses - meaning every vial was fully used - they must add JZ to the claim. If even one drop is thrown away, they can’t use JZ. This sounds minor, but it’s caused major headaches.
Before JZ, providers didn’t need to track discarded amounts. Now, they must document every vial used, note whether any was wasted, and code it correctly. A 2023 survey of gastroenterology practices found a 30% spike in billing staff time spent just on this one modifier. One clinic reported going from 10 minutes per claim to 13 minutes - and they’re still making mistakes. Claim denials for missing or incorrect JZ codes jumped 40% in the first six months after implementation.
The rule was meant to prevent overbilling - where providers bill for more than they used - but it created a new administrative burden. Many providers now use barcode scanners and electronic records to track vial usage, but not all clinics have the tech. Smaller practices are still relying on paper logs and manual checks, which leads to errors.

Why Biosimilar Adoption Is Still Slow in the U.S.
The U.S. has 32 FDA-approved biosimilars as of late 2023. But market share? Only about 35% for mature products like infliximab biosimilars. In Europe, where pricing is more aggressive and reimbursement is based on the cheapest option, adoption hits 75-85% in the same time frame.
Why the gap? It’s not just about price. It’s about how much providers get paid. In Germany or the UK, a hospital gets paid the same amount whether they use the brand or the biosimilar - so they choose the cheaper one. In the U.S., providers still earn more from the brand because of the 6% add-on tied to the reference product’s price. A 2022 study by the Community Oncology Alliance found that 68% of cancer clinics initially confused the new coding system, and 42% had claims denied in the first six months after 2018. Even today, 87% of providers say they still prefer the reference product for high-revenue drugs.
Manufacturers are trying to help. Fresenius Kabi, maker of STIMUFEND®, created a free billing guide with updated codes, modifiers, and examples. Over 87% of providers who used it said it reduced errors. But CMS’s own documentation? Only 58% of providers say it’s enough to solve real-world billing problems.
What’s Next? The Fight Over the 6% Add-On
MedPAC - the Medicare Payment Advisory Commission - has been pushing for change since 2021. Their proposal? Scrap the 6% add-on based on the reference product. Instead, pay biosimilars 106% of their own ASP, and maybe even lower the add-on to 5% or 4%. Some experts suggest a fixed-dollar add-on - say $100 per dose - so everyone gets the same extra payment regardless of price.
Avalere Health estimated that if CMS eliminated the reference product’s ASP from the biosimilar add-on, utilization could jump 15-20 percentage points. That’s billions in savings. The Congressional Budget Office says a full switch to “least costly alternative” payment - where Medicare pays 106% of the average price across all biosimilars and the brand - could save $3.2 billion over 10 years.
But there’s a catch. If providers earn less from biosimilars, manufacturers might cut prices even further to compete. That could hurt innovation. The industry argues that if margins shrink too much, new biosimilars won’t get developed. It’s a balancing act: encourage savings without killing future competition.

What Providers Need to Do Right Now
If you’re a clinic, hospital, or pharmacy billing for biosimilars, here’s what you must do:
- Use the correct HCPCS code for the exact biosimilar administered. Don’t guess. Check CMS’s quarterly updates.
- For infliximab and other drugs with the JZ modifier, track discarded vials. If you use every drop, add JZ. If not, leave it off.
- Verify payment rates with the latest Medicare Physician Fee Schedule. Rates change every July 1 and October 1.
- Train your billing staff. The 2018 transition required 40-60 hours of training per practice. Don’t assume your team knows the rules.
- Use manufacturer guides. Fresenius, Sandoz, and Amgen all publish free, updated coding handbooks. Use them.
Best practices? Implement a dual-check system. Have the pharmacist confirm the product and code before the bill goes out. One oncology center cut billing errors from 15% to under 3% using this method. That’s not just money saved - it’s fewer denied claims, fewer patient complaints, and less staff stress.
Final Reality Check
Biosimilars are not generics. They’re not cheaper versions of the same chemical. They’re complex biological products with their own manufacturing processes, pricing, and billing codes. The current system was designed to help them enter the market - and it did. But it wasn’t designed to make them the default choice.
Right now, the system rewards the status quo. The brand-name drug still gets paid more per dose than its biosimilar counterpart, even if the biosimilar is 20% cheaper. That’s why adoption lags. Until CMS changes how it calculates the 6% add-on, providers will keep prescribing the more expensive option - not because of clinical reasons, but because of billing rules.
The future of biosimilars in the U.S. doesn’t depend on whether they’re safe or effective. It depends on whether the money line up.
Do biosimilars use the same HCPCS code as the original biologic?
No. Before 2018, some biosimilars shared a single code with the reference product. Since January 2018, each FDA-approved biosimilar has its own unique HCPCS code - either a Q-code (temporary) or J-code (permanent). This change was made to ensure accurate reimbursement based on the actual cost of each biosimilar, not a blended average.
Why do providers get paid more for the brand-name biologic than the biosimilar?
Medicare pays 100% of the biosimilar’s ASP plus 6% of the reference product’s ASP. Since the reference product is usually more expensive, the 6% add-on is higher than what the biosimilar’s own ASP would generate. For example, if the reference drug costs $2,500 and the biosimilar costs $2,000, the provider earns $150 in add-on revenue for the brand but only $120 for the biosimilar - a $30 difference per dose. This creates a financial incentive to keep using the more expensive drug.
What is the JZ modifier and why is it required?
The JZ modifier is required for claims involving infliximab and other drugs where no vial was discarded. It tells Medicare that the entire vial was used, so no waste occurred. If any amount was thrown away, the modifier must be omitted. This rule, implemented in July 2023, was meant to prevent overbilling but has added significant documentation work for providers, especially in practices without automated vial tracking systems.
How often do Medicare reimbursement rates for biosimilars change?
Medicare updates biosimilar payment rates quarterly, with major changes typically effective on July 1 and October 1 each year. These updates are published in the Medicare Physician Fee Schedule and reflect the latest Average Selling Price (ASP) data submitted by manufacturers. Providers must check these updates regularly - using outdated codes is one of the top reasons for claim denials.
Can Medicare Advantage plans pay differently than traditional Medicare Part B for biosimilars?
Yes. While traditional Medicare Part B pays 106% of ASP for biosimilars, Medicare Advantage plans are private insurers and can set their own reimbursement rates. Many pay between 100% and 103% of ASP, and some even negotiate lower rates. Providers must verify payment policies with each plan, as this can affect patient cost-sharing and overall revenue.
Is there a chance CMS will change the reimbursement system soon?
Yes. In February 2023, CMS issued an Advanced Notice of Proposed Rulemaking asking for public input on changing the 6% add-on structure. Proposals under review include paying biosimilars 106% of their own ASP (not the reference product’s) or using a fixed-dollar add-on. MedPAC has also recommended a "least costly alternative" model, where Medicare pays 106% of the average price across all biosimilars and the brand. Changes could be implemented by 2025.


 Medications
Medications


Gabrielle Conroy
February 25, 2026 AT 20:10Spenser Bickett
February 26, 2026 AT 15:28Christina VanOsdol
February 28, 2026 AT 14:23Alfred Noble
March 1, 2026 AT 19:29Matthew Brooker
March 3, 2026 AT 18:55Emily Wolff
March 4, 2026 AT 04:08Joseph Cantu
March 4, 2026 AT 05:34Jacob Carthy
March 5, 2026 AT 17:21Lisandra Lautert
March 6, 2026 AT 00:42Cory L
March 7, 2026 AT 03:09Bhaskar Anand
March 8, 2026 AT 05:11William James
March 8, 2026 AT 05:55David McKie
March 9, 2026 AT 08:02Southern Indiana Paleontology Institute
March 9, 2026 AT 13:49Gabrielle Conroy
March 9, 2026 AT 18:15