Author: Lillian Everhart
Learn how to evaluate generic drug coverage when switching health plans. Understand formulary tiers, deductible impacts, and avoid common pitfalls to save money on prescriptions.
Learn how drug take-back programs work to safely dispose of expired medications. Find out where to locate drop boxes, what items are accepted, and why proper disposal protects your family and the environment.
Learn how to access FDA side effect data using OpenFDA and FAERS APIs. This guide covers authentication, query syntax, MedDRA coding, and best practices for detecting safety signals in drug adverse event reports.
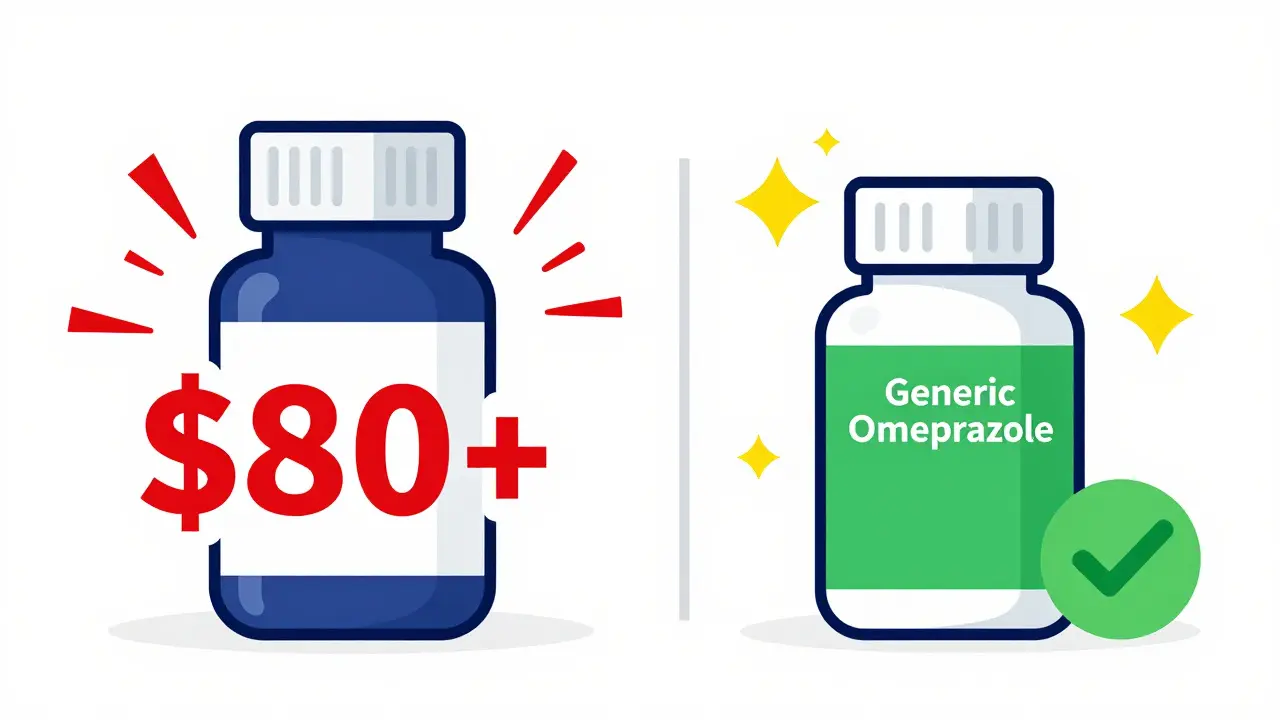
Save money on heartburn relief by buying generic Prilosec (omeprazole) online. Compare prices from top retailers, learn about dosage options, and discover safe purchasing strategies to cut costs by up to 97%.
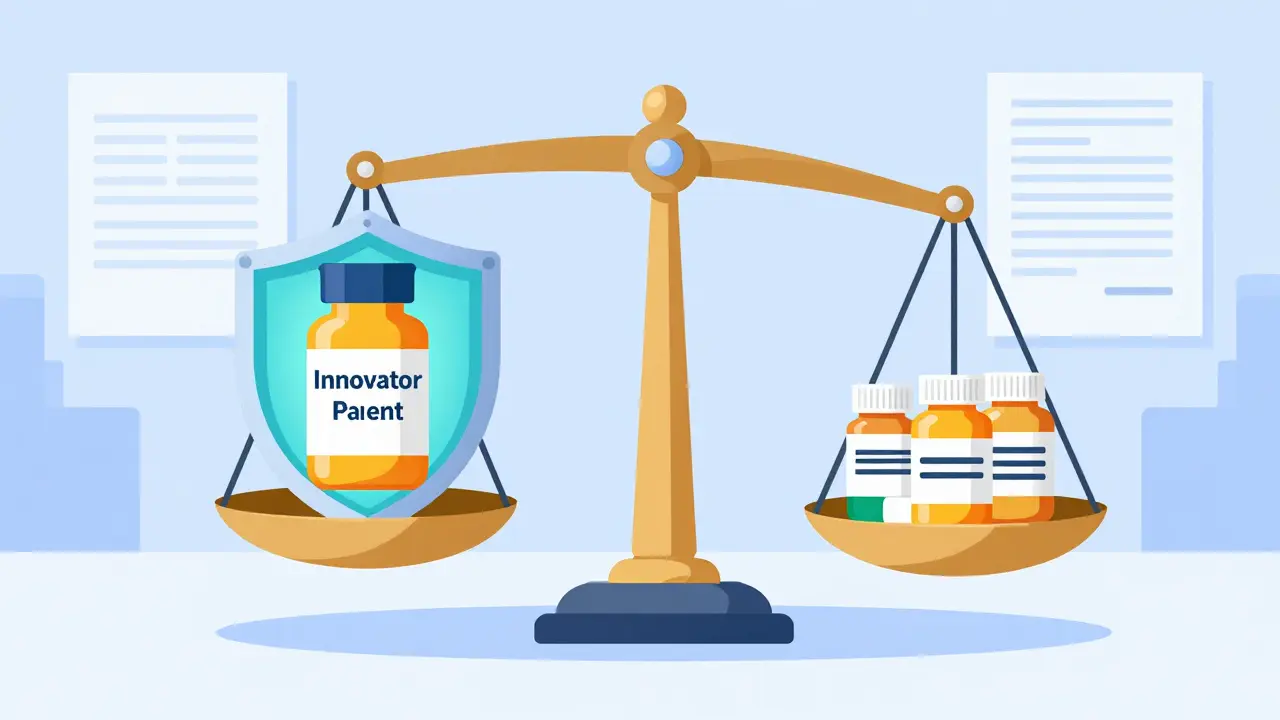
Explore how the FDA approves generic drugs via the ANDA process, ensuring safety and savings. Learn about bioequivalence standards, GDUFA timelines, and the impact of the Hatch-Waxman Act on your medication.
A clear guide on getting vaccinated while on immunosuppressants. Learn the critical differences between live and inactivated vaccines, timing windows for drugs like rituximab and steroids, and specific advice for flu, COVID-19, and shingles protection.
Learn the risks of buying generic Ativan online without a prescription. Discover legal ways to save money on lorazepam using discount coupons and verify safe pharmacies.
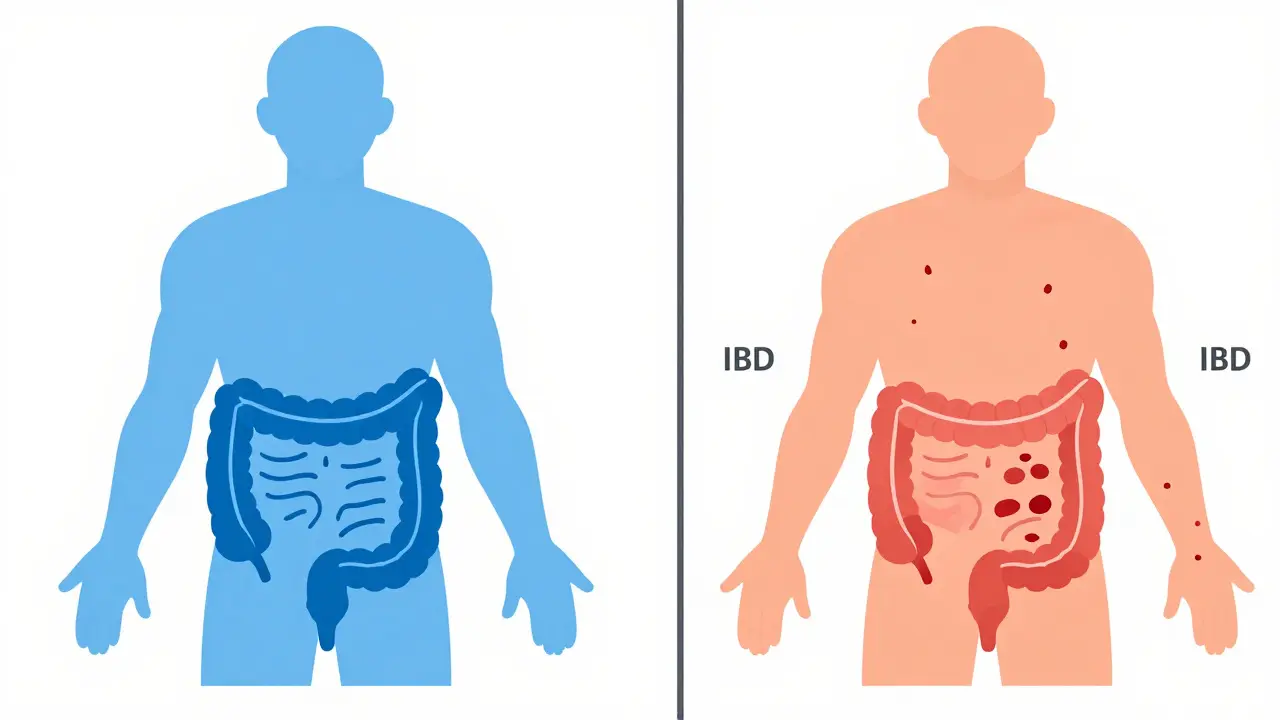
Learn the critical differences between IBS and IBD. We explain how functional disorders differ from inflammatory diseases, covering symptoms, diagnostic tests like fecal calprotectin, and treatment options for better gut health.
A deep dive into the Asian generic pharmaceutical landscape, comparing India's volume dominance with China's API control and the rise of emerging hubs.
Explore why independent Quality Assurance Units are critical for production safety, the risks of integrated structures, and how to maintain regulatory compliance.








 Medications
Medications




