Imagine a factory where the person deciding if a batch of medicine is safe to ship is the same person whose bonus depends on meeting a monthly production quota. It sounds like a recipe for disaster, right? This is exactly why regulatory bodies insist on a strict wall between who makes the product and who checks it. When production pressure hits, the temptation to "just let it slide" can be overwhelming. That's where Quality Assurance Units come in-not as a hurdle to efficiency, but as the essential safety valve that prevents catastrophic failures.
| Key Feature | Requirement | Why It Matters |
|---|---|---|
| Reporting Line | Direct to Executive/Board | Removes production-based pressure |
| Authority | Power to reject batches | Ensures safety over speed |
| Staffing | 8-12% of total workforce | Prevents "rubber stamping" due to fatigue |
What exactly is a Quality Assurance Unit?
In high-stakes industries like pharmaceuticals or nuclear energy, a Quality Assurance Unit (or QU) is a formally designated organizational structure. It isn't just a set of rules; it's a team of people who are legally mandated to stay independent from the production side of the house. According to the FDA, the people developing the product, the people manufacturing it, and the QU must remain separate.
Think of it like a courtroom. You wouldn't want the prosecutor also serving as the judge. In the same vein, the QU acts as the impartial judge of the production process. Their primary job is to ensure that Good Manufacturing Practice (GMP) is followed to the letter. If they find a deviation-even a small one-they have the authority to put a "quality hold" on a batch, regardless of whether the production manager is screaming about a deadline.
The High Cost of Integrated Quality
You might wonder: "Wouldn't it be faster if quality and production worked as one team?" Some experts, like those at MIT, suggest that excessive separation can create communication silos that slow down corrective actions. In some cases, integrated teams resolve deviations about 22% faster. But there's a catch: they also make about 17% more "borderline" compliance decisions. In a nuclear plant or a drug facility, "borderline" can mean a lethal mistake.
The data shows that when the line blurs, risks skyrocket. Between 2020 and 2024, FDA warning letters citing failures in QU independence jumped from 29% to 68%. Why? Because when quality reports to production, batch release decisions become subject to pressure. We've seen real-world examples on forums like r/PharmaEngineering where companies tried to merge these roles during restructuring, only to suffer critical deviations that were ignored just to get product out the door. When you remove the independent check, you aren't increasing efficiency; you're increasing the probability of a recall.
How Independent Oversight Works in Practice
True independence isn't just about having a different office; it's about structural power. A compliant QU needs a clear reporting line that bypasses the production chain entirely. If a QU manager has to ask a production VP for permission to report a failure to the CEO, they aren't independent.
The operational side is usually split into two distinct functions: Quality Control (QC) and Quality Assurance (QA). QC is the "hands-on" part-testing raw materials, checking containers, and sampling the finished product to see if it hits the specifications. QA is the "eyes-on" part-reviewing the procedures, auditing records, and analyzing trends to make sure the system itself isn't broken.
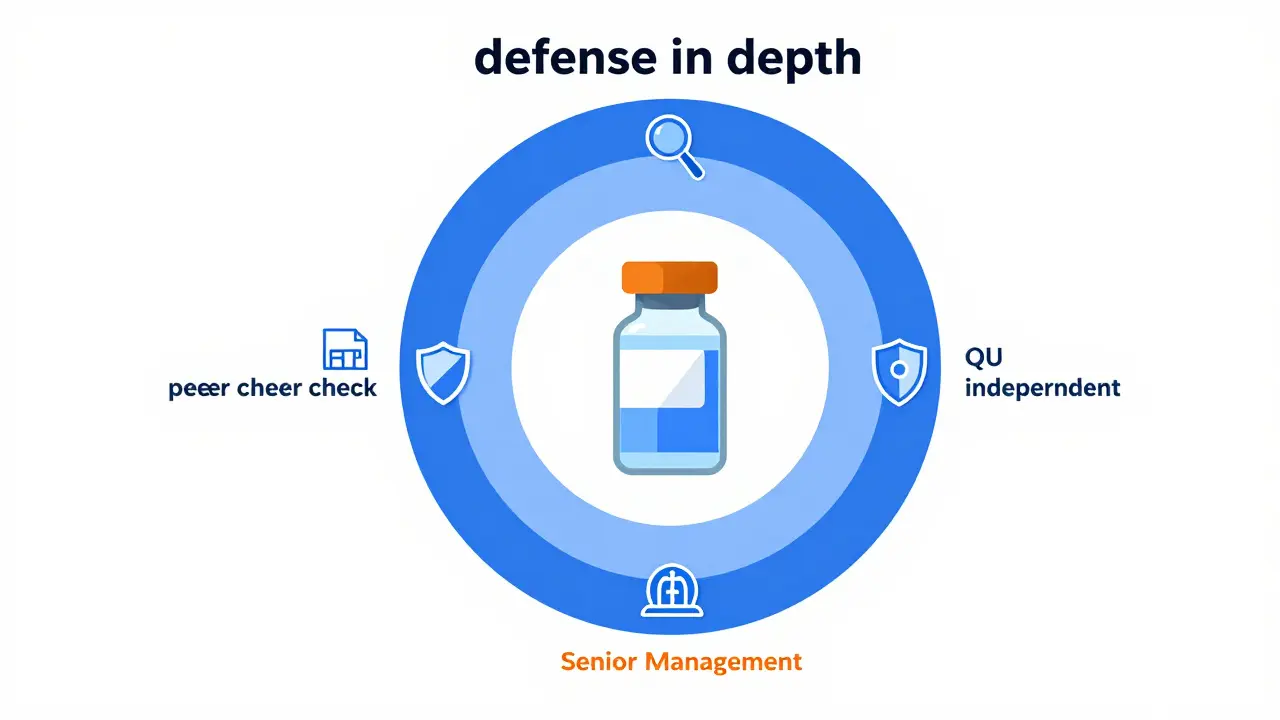
In the nuclear sector, the International Atomic Energy Agency (IAEA) takes this even further with a four-layer approach. They start with peer checking on the line, move to functional oversight by senior managers, then independent oversight, and finally external oversight from regulators. This "defense in depth" strategy has led to a 37% decrease in critical compliance failures compared to organizations with integrated structures.
Common Pitfalls and the "Small Business Trap"
Maintaining a separate QU is expensive. You need dedicated staff, separate budgets, and specialized training. This creates a massive challenge for small companies. In fact, 42% of warning letters for small facilities (under 50 employees) cite failures in QU independence, compared to only 18% for large corporations. In a tiny shop, it's tempting to have one person wear two hats. The FDA allows this only in very rare cases, and only if a second, unrelated person conducts a periodic review of those activities.
Another common failure is the "rubber stamp." When the ratio of QU staff to production staff drops below 1:15, the quality team becomes overwhelmed. Instead of auditing, they start skimming. This leads to a cycle of repeat deviations because the root cause was never actually investigated-the QU was simply too tired or too rushed to care. This is why ISPE benchmarks suggest that QU personnel should make up about 8-12% of the total manufacturing workforce.
Building a Culture of Quality
The biggest hurdle to independence isn't the org chart-it's the culture. Production managers often view the QU as the "police" or a bottleneck. To fix this, some industry leaders have implemented "quality ambassador" programs. For instance, Eli Lilly found that training manufacturing staff in quality principles (while still keeping the reporting lines separate) improved the overall quality culture by 40%.
Success requires a few non-negotiables: 1) QU members must have direct access to the CEO, 2) there must be a documented "quality hold" process that cannot be overridden by production, and 3) staff must be trained in conflict resolution. Since the QU's job is essentially to say "no" to people who want to move fast, they need the soft skills to handle that tension without destroying working relationships.
The Future: AI and Digital Oversight
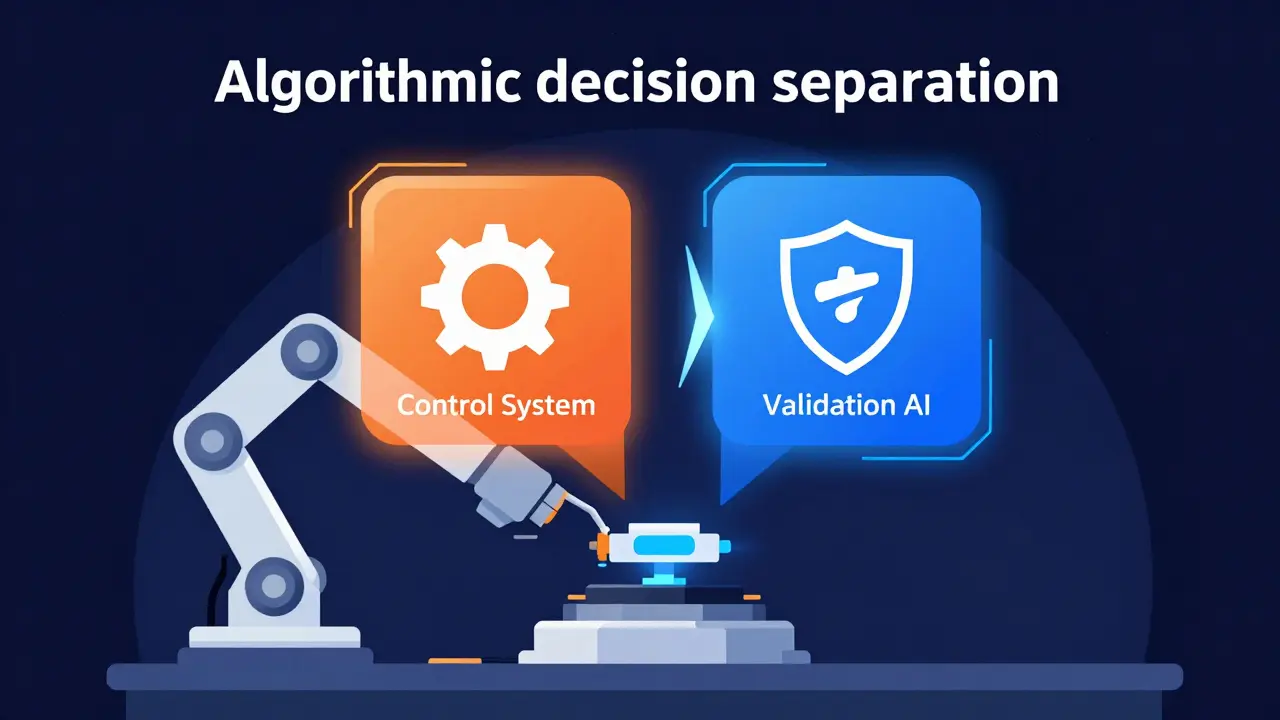
As we move toward 2026 and beyond, the lines are blurring again due to technology. We're seeing the rise of AI-driven production systems where quality checks happen in real-time via sensors and algorithms. The FDA is currently grappling with this in their 2025 draft guidance on digital manufacturing. If an AI makes a real-time decision to reject a part, who is the "independent unit"? Is it the person who wrote the code? The person who validated the model?
The industry is shifting toward "algorithmic decision separation." Instead of just having a different person sign a paper, the independence is built into the software architecture. The system that controls the machine cannot be the same system that approves the quality release. While the tools are changing, the core principle remains: the entity that creates the value cannot be the entity that validates the safety.
Can a production manager also be a quality manager in a small company?
Generally, no. Regulatory bodies like the FDA strongly discourage this because it creates a conflict of interest. If it absolutely must happen due to extreme resource constraints, the FDA requires another qualified individual, who is not involved in production, to conduct a periodic and independent review of all Quality Unit activities to ensure objectivity.
What is the main difference between QA and QC within the Quality Unit?
Quality Control (QC) is focused on the product; it involves testing materials and finished goods to ensure they meet specifications. Quality Assurance (QA) is focused on the process; it involves auditing procedures, reviewing records, and ensuring the entire quality system is functioning correctly to prevent defects before they happen.
How do I know if my Quality Unit is truly independent?
Check your organizational chart. Does the head of the QU report to the Head of Production or the CEO/Board? If they report to production, they aren't independent. Additionally, does the QU have the documented authority to stop a shipment or reject a batch without needing approval from the production manager? If not, your independence is superficial.
Why are FDA warning letters increasing for QU independence failures?
There is a growing trend of companies attempting to lean out their operations by integrating quality and production roles. Regulators have noticed an increase in data integrity violations and "rubber stamping" of records, leading them to crack down more aggressively on facilities that don't maintain a strict organizational divide.
What happens if the QU and Production teams disagree on a batch release?
In a compliant system, the QU's decision is final. The QU has the ultimate authority to approve or reject a batch. While there may be a process for the production team to provide more data or a re-test request, the final "Go/No-Go" decision rests solely with the Quality Unit to ensure that safety is never compromised for the sake of a deadline.



 Medications
Medications




Brittney Prince
April 25, 2026 AT 13:41Yeah right, like the FDA actually cares about our safety and isn't just in the pocket of big pharma to make sure the paperwork looks pretty while they keep shipping the same junk anyway.
Nila Sawyer
April 25, 2026 AT 18:53This is such an inspiring look at how we can make the world safer for everyone by just having a little bit of organized oversight and some really great people in the right places! 🌟 It's so wonderful to see how a structured approach can actually save lives and give us all peace of mind when we're taking our medications, and I truly believe that if we all just support these quality units, we can reach a future where errors are completely non-existent! ✨💖
Beena Garud
April 26, 2026 AT 09:00The dichotomy between efficiency and integrity is a timeless struggle within industrial ethics. One must contemplate whether the pursuit of speed is inherently antithetical to the pursuit of perfection, for when the gaze of the observer is compromised, the truth of the object is lost.
suresh kumar
April 27, 2026 AT 06:26Man, some of these corporate org charts are just a giant game of musical chairs where everyone is pretending to be the boss while the actual work is a total dumpster fire of chaos and spicy mistakes!
Majestic Blue Band
April 29, 2026 AT 04:31It's absolutely laughable that we're supposed to trust these so-called "independent" units when they're all paid by the same corporate machine that profits from the very failures they're supposed to prevent, and honestly, this whole push toward AI oversight is just a convenient way for the elites to erase human accountability entirely so that when a disaster inevitably happens, they can just blame a glitch in the code and move on to the next scam while we all suffer the consequences of their negligence.
Karyn Tindall
April 29, 2026 AT 20:31The thought of a "borderline" decision in a nuclear plant is absolutely horrifying! I can't even imagine the sheer terror of knowing someone just "let it slide" to meet a deadline!
sachin singh
April 30, 2026 AT 05:11The mention of algorithmic decision separation is quite fascinating. It seems that the evolution of quality assurance will rely heavily on the robustness of the validation process for these AI models to ensure the independence is truly systemic.