Archive: 2026/02 - Page 2
Bioequivalence standards for generic drugs have long ignored batch-to-batch variability, risking false conclusions. New guidelines now require multi-batch testing to ensure real-world equivalence and patient safety.
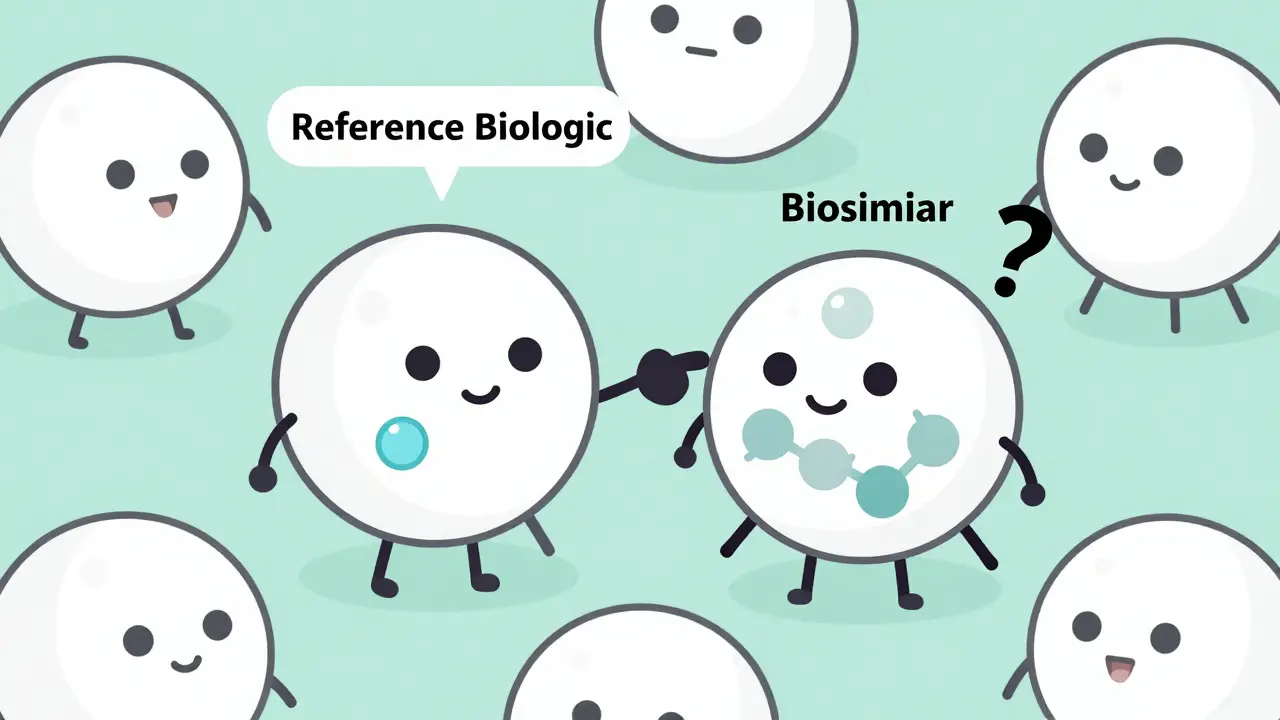
Biosimilars aren't exact copies of biologics - tiny manufacturing differences can trigger immune responses. Learn why some patients develop antibodies, how administration and genetics affect risk, and what real-world data really shows.


 Medications
Medications




